Our paper on a method for obtaining parameters to allow charge-regulation modeling of natural and undefined surfaces has been accepted for publication in Langmuir.
Citation
Brown, D.G., Zhu, H., Albert, L.S., Fox, J.T. 2019. “Rapid characterization and modeling of natural and undefined charge-regulated surfaces in aqueous systems.” Accepted for publication in Langmuir. http://dx.doi.org/10.1021/acs.langmuir.9b02265
Abstract
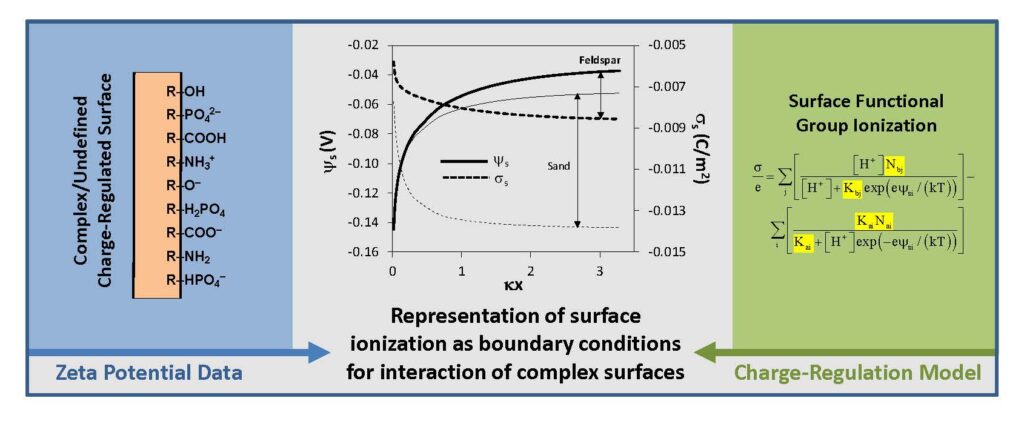
The surfaces of most materials in aqueous systems are charged due to the ionization of surface functional groups. When these surfaces interact, the surface charge, electrostatic potential, and pH will vary as a function of separation distance, and this process is termed the charge-regulation effect. Charge regulation is a controlling factor in the adhesion and transport of colloids and microorganisms in aqueous systems and its modeling requires representation of the pH-charge response of the surfaces, typically provided as the equilibrium constants (K) and site densities (N) of the dominant surface functional groups. Existing methods for obtaining these parameters demonstrate shortcomings when applied to many natural and man-made materials, such as weathered materials, materials with undefined or complex surface structures, and permeable materials, and for materials that don’t provide the requisite high surface area in suspension due to small sample sizes. This hinders inclusion of the charge-regulation effect in colloid and microbial transport studies, and most studies of colloidal and microbial surface interactions use simplifying assumptions – a key example is the routine use of the constant potential assumption in DLVO modeling. Here, we present a robust method that overcomes these issues and provides a rapid means to characterize charge-regulated surfaces using zeta potential data, without requiring a priori knowledge of the material composition. Applying a combined charge-regulation and Gouy-Chapman model, K and N values are obtained that accurately represent the electrostatic response of a charge-regulated surface. This method is demonstrated using activated carbon, aluminum oxide, iron (hydr)oxide, feldspar and silica sand. The resulting K and N values are then used to show the variations in surface charge, electrostatic potential and pH that can occur as these charge-regulated surfaces interact. This method provides a readily-applied experimental approach for characterizing charge-regulated surfaces, with the overall goal to promote the inclusion of charge-regulated interactions into adhesion and transport studies with natural and undefined materials.
